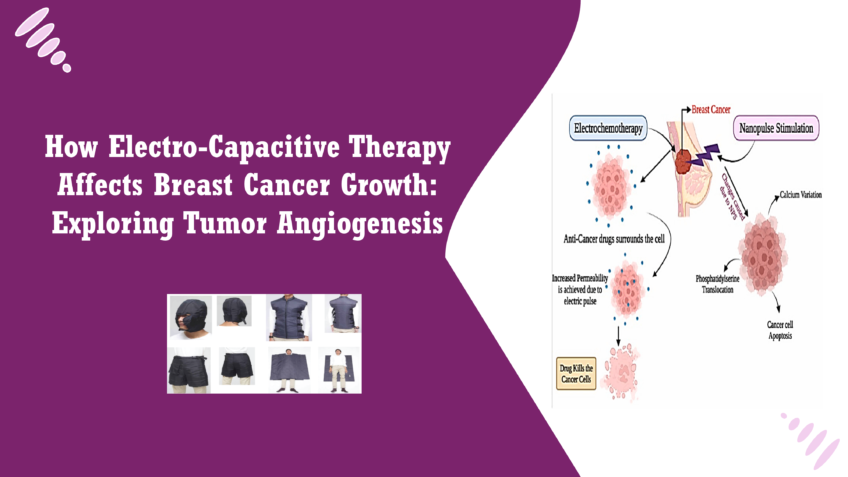
Electro-Capacitive Therapy (ECT) is an innovative medical treatment that employs non-contact alternating electric fields to target and disrupt cancer cell activity. Unlike traditional cancer treatments, which often involve invasive procedures like surgery or systemic therapies such as chemotherapy and radiation, ECT provides a non-invasive approach to cancer care. By using electric fields that can penetrate tissues without direct contact, ECT minimizes physical trauma to the body and reduces the potential for adverse side effects associated with conventional treatments. ECT operates on the principle of capacitive coupling, where electric fields are generated by electrodes placed outside the body. These fields interact with cancer cells, selectively disrupting their normal function, particularly their ability to divide and grow. The non-invasive nature of ECT makes it an appealing option for patients who may not tolerate more aggressive treatments well, allowing for a gentler alternative that aims to reduce cancer progression while preserving the integrity of healthy tissues [1]. How ECT is Used in Cancer Treatment ECT is emerging as a potential adjunct therapy in the treatment of various types of cancer. Its application is based on the understanding that cancer cells often exhibit altered electrical properties compared to normal cells. By harnessing these differences, ECT aims to selectively target and impair cancer cell proliferation without significantly affecting surrounding healthy tissues. In clinical settings, ECT can be administered alongside other treatments, such as chemotherapy or radiation, to enhance their effectiveness while potentially reducing their side effects. The therapy is administered over a specified duration, during which patients are exposed to controlled electric fields that interact with their tumors. This method not only addresses the cancer cells directly but may also influence the tumor microenvironment, thereby impacting factors such as blood vessel formation, known as angiogenesis, which plays a crucial role in tumor growth and metastasis [2]. Why Angiogenesis (Formation of New Blood Vessels) is Important in Cancer Growth Angiogenesis is the physiological process through which new blood vessels form from pre-existing ones. In the context of cancer, angiogenesis is vital for tumor growth and survival. As tumors expand, they require an adequate supply of oxygen and nutrients to sustain their metabolic needs. The process of angiogenesis allows tumors to develop their own blood supply, enabling them to grow larger and potentially spread to other parts of the body (metastasis). Tumors often release specific signaling molecules, such as Vascular Endothelial Growth Factor (VEGF), that stimulate nearby blood vessels to grow towards the tumor. This creates a network of blood vessels that not only nourishes the tumor but also facilitates the dissemination of cancer cells into the bloodstream, leading to metastasis. Targeting angiogenesis has become an important therapeutic strategy in cancer treatment. By inhibiting the formation of new blood vessels, it is possible to starve tumors of the necessary resources they need to grow and spread. Consequently, understanding how ECT affects angiogenesis could provide valuable insights into its role as a therapeutic option in cancer treatment, particularly in reducing tumor growth and improving patient outcomes [3]. Blood vessels play a critical role in supporting the growth and survival of tumors. As tumors develop, they undergo significant metabolic changes that increase their demand for oxygen and nutrients. Without a sufficient blood supply, tumors cannot grow beyond a certain size because they would not receive the necessary elements to support their rapid proliferation. Therefore, the formation of new blood vessels, a process known as angiogenesis, is essential for tumor growth and progression. Blood vessels not only supply the tumor with oxygen and nutrients but also facilitate the removal of waste products generated during cellular metabolism. In cancer, the structure and function of blood vessels can be significantly altered. Tumor-associated blood vessels are often irregular, leaky, and poorly organized, leading to an abnormal distribution of blood flow within the tumor. This aberrant vasculature can create a hypoxic environment, where certain regions of the tumor become oxygen-deficient, driving further angiogenic signaling to recruit more blood vessels [4]. Tumors utilize the process of angiogenesis to secure a constant supply of nutrients and oxygen essential for their growth and survival. As the tumor expands, it secretes various pro-angiogenic factors, the most notable being Vascular Endothelial Growth Factor (VEGF). These factors stimulate surrounding endothelial cells to proliferate and migrate, forming new blood vessels that infiltrate the tumor. Once the new blood vessels are established, they facilitate the transport of oxygen, glucose, and other nutrients into the tumor microenvironment, thereby supporting its growth. Additionally, the newly formed vasculature provides a pathway for cancer cells to enter the bloodstream, allowing them to disseminate and form secondary tumors (metastases) in distant organs. This capacity to adapt and manipulate the blood supply is a hallmark of cancer, making angiogenesis a critical factor in tumor progression [5]. The Significance of Targeting Angiogenesis in Cancer Therapies Given the pivotal role of angiogenesis in tumor growth and metastasis, targeting this process has emerged as a promising therapeutic strategy in cancer treatment. Anti-angiogenic therapies aim to inhibit the formation of new blood vessels, effectively starving the tumor of the nutrients and oxygen it requires to grow. Several approaches have been developed to disrupt angiogenesis. These include the use of monoclonal antibodies that target VEGF (such as Bevacizumab) and small-molecule tyrosine kinase inhibitors that block the signaling pathways involved in angiogenesis. By preventing tumors from establishing a robust blood supply, these therapies can slow tumor growth, reduce the likelihood of metastasis, and potentially enhance the effectiveness of other treatments, such as chemotherapy or radiation. Furthermore, understanding the mechanisms of angiogenesis can help identify biomarkers that predict tumor response to anti-angiogenic therapies, allowing for more personalized and effective treatment strategies. The ability to target angiogenesis not only provides a novel avenue for cancer therapy but also underscores the interconnectedness of the tumor and its microenvironment, highlighting the need for comprehensive treatment approaches that consider both the cancer cells and the supporting structures that enable their growth [6]. How ECT Impacts Tumour Angiogenesis Electro-Capacitive Therapy (ECT) operates by generating alternating electric fields (AEFs) that
Day: November 5, 2024
How a Noncontact Electro Capacitive Cancer Therapy Affects Tumour Growth and Inflammatory Markers
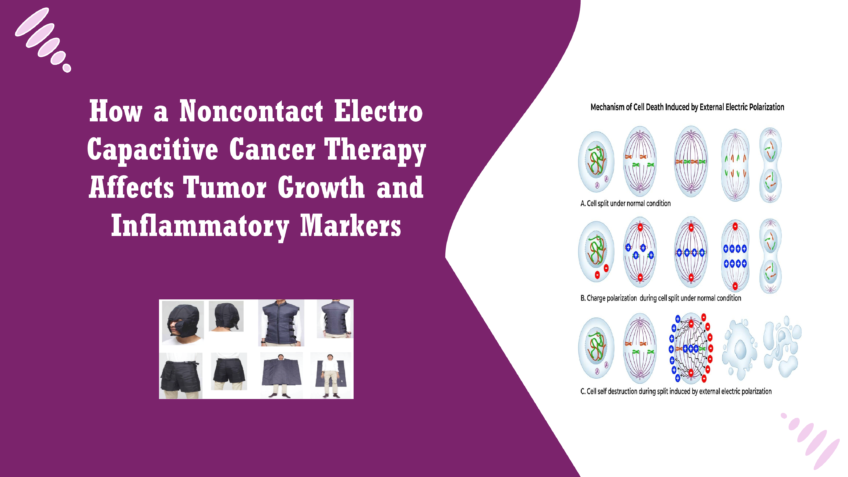
Introduction Cancer remains one of the most pressing health challenges globally, affecting millions of individuals each year. Traditional treatment modalities, including surgery, chemotherapy, and radiation therapy, have been cornerstones in the fight against cancer. However, these approaches often come with significant limitations, such as severe side effects, potential for recurrence, and the development of resistance in cancer cells. Patients frequently experience adverse reactions that can diminish their quality of life, highlighting the urgent need for innovative and less invasive therapeutic options [1]. In response to these challenges, researchers and clinicians are exploring novel therapeutic strategies that harness the body’s biological responses to fight cancer more effectively. One such promising approach is Noncontact Electro Capacitive Cancer Therapy (ECCT). This innovative treatment utilizes low-intensity alternating electric fields to disrupt cancer cell proliferation while minimizing damage to surrounding healthy tissue [2]. Unlike traditional therapies that directly target the tumor, ECCT works from a distance, offering a non-invasive alternative that may reduce the risk of side effects. ECCT has garnered attention for its potential to activate specific cellular pathways that induce apoptosis—programmed cell death—without harming normal cells. Initial studies have shown that this method can significantly hinder tumor growth, making it a subject of interest in preclinical and clinical research [3]. As scientists continue to investigate the molecular mechanisms underlying ECCT, its role in modulating inflammatory markers such as CCL2 and IL18 offers valuable insights into how this therapy could enhance anti-tumor efficacy and reshape the future of cancer treatment. This blog will explore the findings of recent research on ECCT, shedding light on its potential benefits and implications for patients battling cancer [4]. Understanding ECCT Noncontact Electro Capacitive Cancer Therapy (ECCT) operates on the principle of utilizing low-intensity alternating electric fields to disrupt the biological processes of cancer cells. The therapy involves placing capacitive electrodes around the treatment area, creating an electric field that penetrates tissues without direct contact. This non-invasive approach allows for the modulation of cellular activities through the application of specific electric frequencies [5] When cancer cells are exposed to these alternating electric fields, several biological responses can be triggered. The primary mechanism appears to be the induction of apoptosis, a form of programmed cell death that is crucial for eliminating damaged or dysfunctional cells. ECCT has been shown to disrupt the normal mitotic process of cancer cells, causing them to halt their division and ultimately die. This disruption is thought to occur through several pathways, including the destabilization of microtubules during mitosis and the activation of apoptotic signalling cascades[6]. Additionally, ECCT may influence the tumour microenvironment by modulating the activity of surrounding immune cells. Electric fields can impact macrophage function, enhancing their ability to recognize and eliminate cancer cells [7]. This interaction is crucial because a supportive immune response can significantly enhance the efficacy of cancer therapies. The ability of ECCT to induce changes in the expression of inflammatory markers, such as CCL2 and IL18, suggests that this therapy not only targets cancer cells directly but also leverage the immune system to mount a more effective anti-tumour response [8]. Research on ECCT has gained momentum in recent years, particularly due to its potential as a complementary treatment option in oncology. Early studies demonstrated that exposure to low-frequency electric fields could inhibit the growth of various cancer cell lines, including breast cancer, lung cancer, and oral squamous cell carcinoma. For instance, a study found that applying an electric field at 100 kHz effectively reduced the viability of MCF-7 breast cancer cells in vitro, showcasing the therapy’s promise as a non-invasive alternative [9]. In vivo experiments using animal models have also yielded encouraging results. One notable study involving DMBA-induced breast cancer in mice revealed that ECCT significantly reduced tumour size compared to control groups. Researchers observed not only a decrease in tumours mass but also a corresponding increase in apoptotic cells, indicating that the therapy effectively triggered cell death mechanisms [10]. Moreover, investigations into the molecular underpinnings of ECCT have revealed its potential to modulate specific signalling pathways associated with cancer progression. Previous findings suggested that ECCT could upregulate tumour suppressor genes and downregulate oncogenes, contributing to its anti-proliferative effects. The ability of ECCT to affect cytokine expression further highlights its dual action: directly targeting tumour cells while also modifying the inflammatory landscape of the tumour microenvironment [11]. Overall, the growing body of evidence supporting ECCT’s efficacy in cancer treatment underscores its potential to serve as a valuable tool in the oncologist’s arsenal. As research continues, understanding the precise mechanisms through which ECCT operates will be crucial in optimizing its application and integrating it into standard cancer care practices [12]. Mechanism of ECCT In the investigation of Noncontact Electro Capacitive Cancer Therapy (ECCT), researchers employed a well-established animal model using Sprague Dawley rats. This model is widely recognized for its relevance in studying breast cancer due to its similar physiological characteristics to humans. In this study, the rats were induced with tumours using a chemical carcinogen, 7,12-dimethylbenz[a]anthracene (DMBA), which mimics the progression of breast cancer. This approach allowed researchers to create a controlled environment to observe the effects of ECCT on tumour growth and associated inflammatory responses [13]. The experimental design involved four distinct treatment groups, each consisting of six rats. Two groups were DMBA-induced, with one receiving ECCT treatment and the other serving as a control. The remaining two groups were non-DMBA-induced rats, also divided between those receiving ECCT and those not. This comprehensive setup enabled the team to assess the effects of ECCT both on induced tumours and in a healthy context, providing valuable insights into the therapy’s effectiveness [14]. To measure tumour growth and inflammation, researchers employed several advanced techniques, including histological examination and immunohistochemistry. Tumour tissues were collected and processed for staining, which allowed for detailed visualization of cellular structures and the presence of specific proteins [14]. Immunohistochemistry, in particular, was instrumental in assessing the expression levels of markers associated with cell proliferation and apoptosis, such as PCNA (proliferating cell nuclear antigen) and Caspase3. The study’s
Evaluating the Impact of Electro-Capacitive Therapy on Cancer Patient Survival: Insights from Cox Model Analysis
Electro-Capacitive Cancer Therapy (ECCT) is a non-invasive treatment modality designed to target cancer cells through the application of alternating electric fields. By utilizing capacitive coupling, ECCT generates electric fields that penetrate tissue and influence cellular behavior without the need for direct contact. This innovative therapy aims to disrupt the proliferation and survival of cancer cells while minimizing damage to surrounding healthy tissues, thereby reducing the side effects commonly associated with traditional cancer treatments like chemotherapy and radiation. ECCT has garnered significant attention in the medical community, particularly due to its unique mechanism of action. By altering the electrical environment within the tumor microenvironment, ECCT can potentially interfere with key cellular processes, such as cell division and metabolism, thereby impairing the tumor’s ability to grow and spread. The treatment is particularly relevant for patients seeking alternative or complementary therapies alongside conventional cancer treatments, offering a novel approach to enhancing therapeutic outcomes and improving quality of life [1]. Prevalence of Cancer and the Need for Innovative Therapies Cancer remains one of the leading causes of morbidity and mortality worldwide. According to the World Health Organization (WHO), an estimated 10 million deaths were attributed to cancer in 2020, highlighting the pressing need for effective treatment options. The complexity of cancer, characterized by its heterogeneity and the ability to develop resistance to therapies, underscores the urgency for innovative approaches that can provide better outcomes for patients. Despite advancements in cancer treatments over the past few decades, many patients still experience limited efficacy, significant side effects, and challenges with treatment adherence. This necessitates the exploration of novel therapies that can address these shortcomings. ECCT represents a promising addition to the oncology arsenal, as it offers a non-invasive treatment option that may complement existing modalities while targeting tumor growth and enhancing patient outcomes [2]. Significance of Survival Analysis in Evaluating Treatment Efficacy Survival analysis is a statistical approach used to assess the time until an event of interest occurs, such as death or disease progression. In the context of cancer treatment, survival analysis plays a crucial role in evaluating the efficacy of therapeutic interventions, enabling researchers and clinicians to understand how different factors influence patient outcomes. The Cox Proportional Hazards model is one of the most widely used methods in survival analysis, allowing researchers to estimate the effect of various prognostic factors on survival time while accounting for other variables. This model is particularly valuable in oncology, as it provides insights into how treatments, demographics, and clinical characteristics impact the survival of cancer patients. By employing survival analysis, researchers can identify which factors are associated with improved or diminished survival rates, ultimately guiding clinical decision-making and treatment strategies. In the case of ECCT, evaluating its impact on survival outcomes through rigorous statistical analysis helps validate its effectiveness and informs the ongoing development of treatment protocols. Understanding the nuances of survival analysis is therefore essential for comprehensively assessing the potential benefits of ECCT and integrating it into standard cancer care [3]. Survival analysis is a statistical technique that focuses on analyzing the time until an event of interest occurs, such as death, disease progression, or recurrence of cancer. This type of analysis is particularly important in medical research, as it provides insights into the effectiveness of treatments, the natural history of diseases, and the impact of various patient characteristics on health outcomes. In the context of cancer, survival analysis allows researchers to estimate survival probabilities and identify factors that influence patient longevity. By examining the duration of time patients survive after diagnosis or treatment initiation, clinicians can better understand the efficacy of different therapeutic approaches and make more informed decisions regarding patient care. Moreover, survival analysis helps in identifying subgroups of patients who may benefit from specific treatments, thereby contributing to personalized medicine. The ability to account for censored data—patients who have not yet experienced the event of interest or have been lost to follow-up—is one of the key strengths of survival analysis. This feature enables researchers to make robust estimates of survival probabilities, even in cases where complete data is not available, enhancing the reliability of conclusions drawn from clinical studies [4]. Cox Proportional Hazards Model and How It Assesses Risk Factors The Cox Proportional Hazards model is one of the most widely used statistical methods in survival analysis, particularly in oncology. Developed by Sir David Cox in 1972, this semi-parametric model estimates the hazard function, which represents the instantaneous risk of the event occurring at a given time, based on one or more predictor variables. One of the key features of the Cox model is its ability to evaluate the effect of various prognostic factors on survival while controlling for confounding variables. The model generates hazard ratios (HR), which indicate the relative risk of the event occurring for different levels of the predictor variables. For instance, an HR greater than 1 suggests an increased risk associated with a specific factor, while an HR less than 1 indicates a protective effect. The assumption of proportional hazards is fundamental to the Cox model, meaning that the relationship between the predictors and the hazard is constant over time. This characteristic allows researchers to identify significant risk factors in a straightforward manner, making the Cox model a valuable tool in clinical research, particularly when comparing the survival rates of different patient groups or treatment modalities [5]. Prognostic Factors Relevant to Cancer Treatment Prognostic factors are characteristics or variables that can influence the outcome of a disease and the response to treatment. In cancer research, identifying and understanding these factors is crucial for optimizing patient management and improving treatment strategies. Some common prognostic factors in cancer treatment include: Patient Age: Age can significantly impact a patient’s overall health and treatment tolerance, affecting survival outcomes. Tumor Stage: The extent of cancer at diagnosis, often classified using the TNM (Tumor, Node, Metastasis) staging system, is a critical factor influencing prognosis. Histological Grade: The differentiation of cancer cells compared to normal cells can indicate how aggressive the tumor may be. Treatment Type:
Evaluating Survival Factors in Cancer Patients Treated with Electro-Capacitive Therapy: Insights from Weibull Regression Analysis
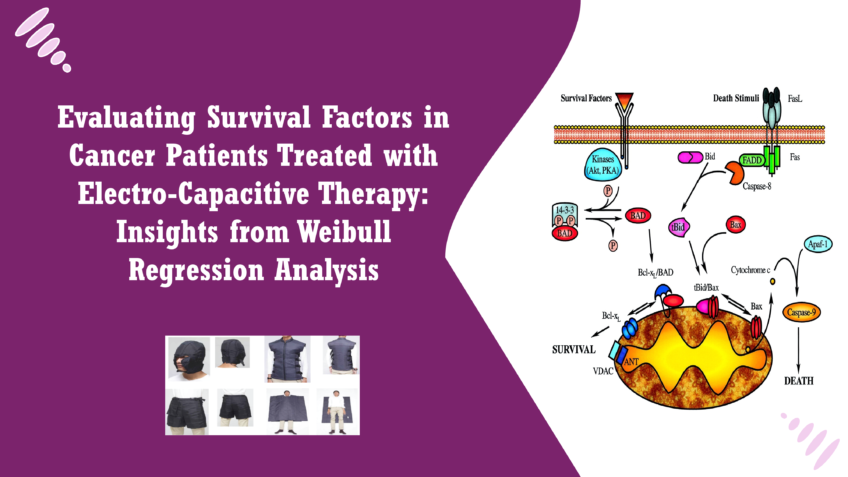
Electro-Capacitive Cancer Therapy (ECCT) is a non-invasive treatment modality designed to target and inhibit the growth of cancer cells. This innovative therapy utilizes alternating electric fields generated through a specialized device to disrupt the cellular processes that facilitate tumor growth. By applying controlled electric fields, ECCT aims to induce apoptosis (programmed cell death) in cancer cells while preserving surrounding healthy tissues, thus minimizing the side effects often associated with traditional cancer treatments such as chemotherapy and radiation therapy. The underlying principle of ECCT is based on the interaction between electric fields and biological tissues. Research suggests that electric fields can alter the cellular membrane potential and influence cellular activities such as proliferation and migration. As a result, ECCT represents a promising therapeutic approach for various types of cancers, potentially improving patient outcomes and quality of life [1]. Cancer remains one of the leading causes of morbidity and mortality worldwide, with approximately 19.3 million new cases and nearly 10 million cancer-related deaths reported in 2020 alone, according to the World Health Organization (WHO). The incidence of cancer is expected to rise, particularly in developing countries where access to advanced medical care and treatment options may be limited. This growing cancer burden highlights the urgent need for innovative therapeutic approaches that are not only effective but also accessible and minimally invasive. The limitations of traditional cancer treatments, including chemotherapy, radiation, and surgery, often lead to severe side effects and a significant impact on patients’ quality of life. As a result, there is a critical demand for novel therapies that can enhance the effectiveness of existing treatments while reducing adverse effects. ECCT represents one such approach, harnessing the power of electrical fields to offer a complementary option in the fight against cancer [2]. Survival analysis is a statistical method used to evaluate the time until an event of interest occurs, commonly referred to in cancer research as the time until death or disease progression. It provides valuable insights into the effectiveness of treatments and helps identify factors that influence patient outcomes. By analyzing survival data, researchers can assess the efficacy of therapies, compare different treatment modalities, and identify prognostic factors that affect survival rates. In the context of ECCT, survival analysis plays a pivotal role in determining the therapy’s impact on patient survival and identifying which factors contribute to improved outcomes. By employing models such as the Weibull regression model, researchers can evaluate the relationship between various prognostic factors—including treatment frequency, patient demographics, and cancer type—and survival rates. This information is crucial for optimizing treatment protocols and personalizing care for cancer patients, ultimately enhancing their chances of survival and quality of life. Overall, understanding ECCT and its implications in the broader context of cancer treatment underscores the importance of innovative approaches and the necessity of rigorous evaluation methods such as survival analysis to establish their efficacy and safety in clinical practice. Survival analysis is a statistical approach used to analyze the time until a specific event occurs, such as death or disease progression. In the field of cancer research, survival analysis is critical for assessing the effectiveness of treatments and understanding the prognosis of patients. It allows researchers to estimate survival probabilities and identify factors that may influence patient outcomes over time [3]. The primary goals of survival analysis are to: 1. Estimate Survival Times: By analyzing data from patient cohorts, researchers can estimate the median survival time and survival rates at different time intervals, helping to understand the effectiveness of various treatment modalities. Compare Treatment Efficacy: Survival analysis enables the comparison of different treatment options, providing insights into which therapies yield better outcomes for specific cancer types or patient populations. Identify Prognostic Factors: By evaluating various factors—such as age, tumor stage, and treatment frequency—researchers can identify key prognostic indicators that significantly affect survival. Understanding these factors is essential for personalizing treatment strategies and improving patient management. Survival analysis techniques commonly employed in medical research include Kaplan-Meier estimation, log-rank tests for comparing survival curves, and regression models that account for covariates affecting survival [4]. Weibull Regression Model and Its Application in Evaluating Survival Factors The Weibull regression model is a powerful statistical tool frequently used in survival analysis, particularly for modeling time-to-event data in the presence of censoring. The model is characterized by its flexibility in accommodating different hazard functions, making it suitable for various survival data scenarios. Key features of the Weibull regression model include: Hazard Function: The model allows for the estimation of hazard rates, which represent the risk of the event occurring at a specific time point, conditional on survival until that time. Shape Parameter: The Weibull model includes a shape parameter that indicates the nature of the hazard function—whether it is increasing, constant, or decreasing over time. This feature is crucial for accurately modeling survival data from cancer patients with varying prognosis. Prognostic Factor Assessment: The Weibull regression model can incorporate multiple covariates (e.g., age, treatment frequency) to evaluate their effects on survival. This allows researchers to identify significant factors influencing patient outcomes. In the context of Electro-Capacitive Cancer Therapy (ECCT), the Weibull regression model can help assess the impact of various treatment-related factors on patient survival, providing insights that can guide clinical decision-making and improve therapeutic strategies. [5] Identifying prognostic factors is critical in cancer treatment for several reasons: 1. Personalized Treatment: Understanding the factors that influence survival allows for tailored treatment plans based on individual patient characteristics, thereby enhancing the likelihood of positive outcomes. Resource Allocation: By recognizing high-risk patients, healthcare providers can allocate resources more efficiently, ensuring that patients who require more intensive monitoring or alternative treatments receive appropriate care. Clinical Decision-Making: Prognostic factors provide essential information for clinicians when making treatment decisions. For example, if a particular factor is associated with poor outcomes, clinicians may consider alternative therapies or more aggressive treatment approaches. Research and Development: Identifying and validating prognostic factors contributes to ongoing research efforts aimed at developing more effective treatment modalities and improving patient survival rates. Research on ECCT has identified several