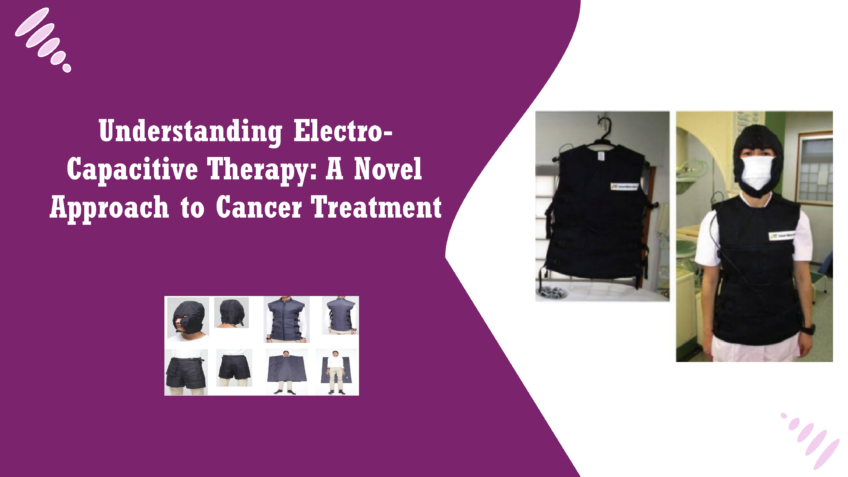
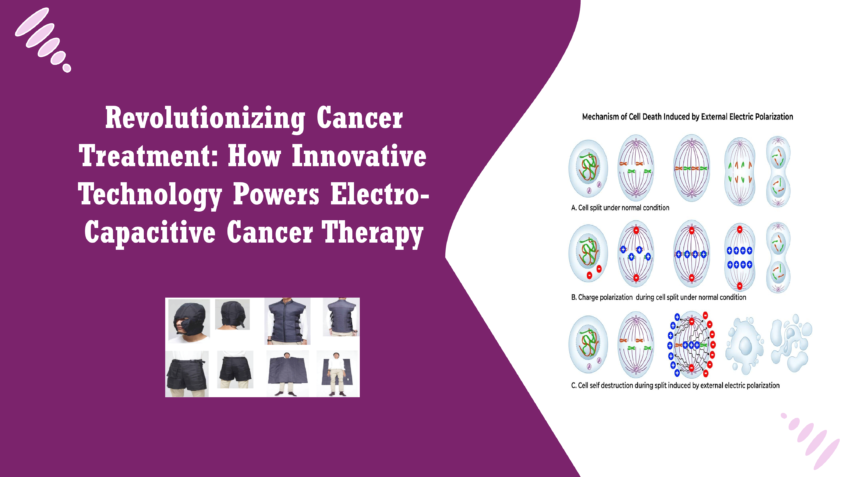
What is Electro-Capacitive Therapy? Electro-Capacitive Therapy (ECT) is a non-invasive therapeutic technique that leverages electrical fields to treat various diseases, with a growing focus on its application in cancer treatment. It involves the use of capacitive electrical fields to disrupt cellular activity, particularly targeting cancer cells while sparing healthy tissues. ECT works by placing electrodes on the body, which emit low-level electrical pulses designed to interact with cells at the molecular level. This interaction has shown the potential to inhibit cancer cell growth and promote their destruction [1]. How ECT is emerging as a novel cancer treatment In the realm of oncology, Electro-Capacitive Therapy is gaining attention as an innovative approach to tackling cancer. Traditional cancer treatments like chemotherapy, radiation, and surgery can have significant side effects, often harming healthy cells in the process. ECT offers a potentially safer alternative by selectively disrupting cancer cell function. Early studies and clinical trials suggest that ECT may enhance the precision of cancer treatment, reduce damage to healthy tissue, and lessen the side effects associated with traditional therapies. Because of this, ECT is increasingly being explored as part of a multimodal treatment strategy, potentially used in conjunction with existing therapies [2]. The science behind using electric fields in medical therapies The use of electric fields in medical treatments is not a new concept. For decades, electric fields have been used to promote tissue regeneration, wound healing, and pain management. In the context of cancer, electric fields exert their effects by influencing the electrical properties of cells. Cancer cells, due to their high rate of division, are particularly sensitive to external electric fields. ECT harnesses this sensitivity by applying low-intensity, alternating electric fields that interfere with key cellular processes such as mitosis (cell division). This disruption not only prevents the proliferation of cancer cells but may also lead to their destruction, through mechanisms like apoptosis (programmed cell death), making ECT a promising addition to the arsenal of cancer therapies [3]. This novel approach stems from a deeper understanding of bioelectricity—how cells communicate and function using electrical signals. As research continues to uncover the nuances of ECT’s mechanisms, its role in the future of cancer therapy looks increasingly promising. How Does Electro-Capacitive Therapy Work? Electro-Capacitive Therapy (ECT) operates on the principle of capacitive electrical fields, which are formed between two conductive objects (electrodes) that create a non-contact interaction with biological tissues. When an alternating electric current is applied between these electrodes, it generates an oscillating electric field that interacts with the cells in the tissue, without penetrating the skin directly. This is a key aspect of capacitive coupling—where energy is transferred without the need for direct electrical conduction. In a biological context, capacitive fields influence cells by altering the distribution of electrical charges on their membranes. Cells, especially rapidly dividing ones like cancer cells, are highly sensitive to these external electrical environments. The oscillating electric fields in ECT can modify cellular functions by disrupting the normal electrochemical gradients across cell membranes, affecting the stability and functionality of cancer cells [4]. Cancer cells differ from normal cells in that they proliferate uncontrollably and have a unique cellular environment. One of the critical stages of cancer progression is mitosis, where cancer cells divide and multiply at an accelerated rate. ECT leverages the sensitivity of these dividing cells to electric fields. When cancer cells attempt to undergo mitosis, the applied capacitive electrical fields interfere with their ability to align and segregate chromosomes properly. This disruption can cause significant structural stress within the cancer cells, leading to multiple outcomes: the cell cycle may be halted (inhibiting further cell division), or the cells may undergo apoptosis (programmed cell death) because of the inability to maintain normal cellular processes. Essentially, the capacitive fields “confuse” the cancer cells’ internal signaling and regulatory mechanisms, leading to their destruction or preventing their growth. How ECT targets cancer cells without harming healthy cells One of the remarkable features of Electro-Capacitive Therapy is its ability to target cancer cells specifically, while minimizing damage to healthy tissue. This selectivity arises from two main factors: the nature of cancer cells and the frequency of the applied electric fields. Cancer cells are particularly vulnerable to ECT because they exhibit a high rate of division and have abnormal membrane properties. Their rapid mitotic activity makes them more responsive to the effects of external electric fields, which disrupt the mitosis process. In contrast, most healthy cells divide at a much slower rate, and their normal membrane potential allows them to be less affected by these external fields. Moreover, the parameters of ECT—such as the frequency and intensity of the electric fields—are optimized to specifically disrupt the behavior of cancer cells. Healthy cells, which are in a more stable, non-dividing state, do not experience the same level of stress from the electric fields, allowing them to continue functioning normally. This selectivity is a key advantage of ECT, as it minimizes the collateral damage typically associated with traditional cancer treatments like chemotherapy and radiation, which can harm both healthy and cancerous cells. In summary, ECT works by creating electrical fields that selectively interfere with the abnormal processes in cancer cells while sparing healthy cells, making it a highly targeted and potentially safer cancer treatment option [5]. Mechanisms of Action in Cancer Treatment Mitosis, the process of cell division, is a key target in Electro-Capacitive Therapy (ECT). Cancer cells are characterized by their rapid and uncontrolled division, making them particularly susceptible to interventions that disrupt mitosis. During mitosis, cells go through a series of well-coordinated phases that involve DNA replication, chromosome alignment, and eventual division into two daughter cells. For this process to occur smoothly, cancer cells must maintain proper electrical and structural integrity. ECT interferes with mitosis by applying alternating electrical fields that disrupt the proper alignment and separation of chromosomes. Specifically, ECT affects the mitotic spindle—a structure essential for guiding chromosomes during cell division. The capacitive electrical fields generated in ECT cause physical stress on the spindle’s microtubules, preventing chromosomes from
Day: November 5, 2024
Optimizing Electro-Capacitive Cancer Therapy: The Role of Electric Field Distribution in Treatment Efficacy
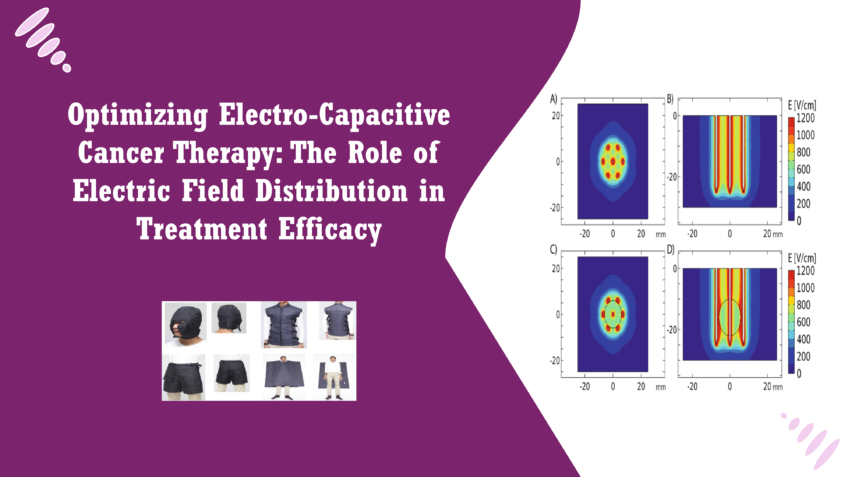
Electro-Capacitive Cancer Therapy (ECCT) is an innovative and non-invasive cancer treatment that leverages the effects of electric fields (EF) on cellular activity. ECCT specifically targets the electrical characteristics of cancer cells, which differ from those of healthy cells. Cancer cells exhibit abnormal electrical behaviour, especially during mitosis (cell division), where their electrical properties can be exploited to disrupt cell proliferation. ECCT delivers carefully controlled electric fields at intermediate frequencies to selectively interfere with cancer cell division while sparing normal cells, making it a promising approach in cancer treatment. Unlike conventional methods like chemotherapy and radiation, which often affect both cancerous and healthy tissues, ECCT offers a more targeted therapy with potentially fewer side effects. The importance of optimizing ECCT for effective treatment lies in its ability to maximize the impact on cancer cells while minimizing unintended effects on surrounding healthy tissue. The efficacy of ECCT depends on how accurately the electric fields are applied to the tumor site. Variables such as tumor size, location, and the properties of the surrounding tissue can significantly influence treatment outcomes. Without precise control and optimization, electric fields may not reach the necessary intensity to disrupt cancer cells or may affect healthy tissues, reducing the overall effectiveness of the treatment. Therefore, optimizing ECCT involves refining treatment parameters to ensure that electric field intensity is distributed in a way that maximally impacts cancer cells while sparing normal tissue [1]. This blog will focus on understanding how electric field (EF) distribution affects treatment outcomes in ECCT. By examining how EF interacts with different types of tissue and the role of dielectric properties (how materials respond to electric fields), researchers can improve the design and application of ECCT. In particular, this article will explore the use of Wire Mesh Tomography (WMT) to measure EF distribution in various media, offering insights into how this technique can help refine and optimize ECCT for more effective cancer treatment. Understanding EF distribution is crucial because it not only enhances treatment accuracy but also ensures that cancer cells receive the maximum therapeutic benefit, leading to improved patient outcomes. Electro-Capacitive Cancer Therapy (ECCT) is based on the principle that cancer cells exhibit distinct electrical properties compared to normal, healthy cells. ECCT harnesses this difference by using externally applied electric fields (EF) to inhibit cancer cell growth and proliferation. In essence, ECCT generates electric fields that interfere with the abnormal electrical activity of cancer cells, especially during critical stages like mitosis, when cells are actively dividing [2]. The electric fields used in ECCT are typically tuned to intermediate frequencies, which have been shown to disrupt key cellular processes in cancer cells without significantly affecting the surrounding normal cells. The mechanism by which ECCT inhibits cancer cell growth involves the interaction between electric fields and the biophysical characteristics of the cells. Cancer cells, due to their uncontrolled growth and division, have altered membrane potentials, ion channel activity, and overall electrical conductivity compared to normal cells. These differences in electrical properties make cancer cells more susceptible to disruption by externally applied electric fields. In ECCT, the electric fields target the cancer cells’ abnormal activity by interfering with their mitotic spindle formation during cell division. Mitosis is a highly regulated process, and any disruption to the spindle apparatus can lead to failed division or even cell death. By applying electric fields at specific intensities and frequencies, ECCT selectively affects cancer cells’ ability to divide, effectively halting tumor growth. Importantly, healthy cells, which maintain more stable electrical properties and undergo less frequent division, are far less impacted by these fields, making ECCT a selective and targeted therapy [3]. One of the key advantages of ECCT is this ability to exploit cancer cells’ vulnerabilities without causing significant damage to normal tissues. Unlike chemotherapy or radiation, which damage both healthy and cancerous tissues, ECCT focuses primarily on cells that display abnormal electrical behavior. This selectivity not only enhances the treatment’s effectiveness but also reduces side effects, offering a less invasive alternative to conventional cancer treatments. Moreover, ECCT has been studied for its potential to work synergistically with other treatments, potentially improving patient outcomes across a variety of cancer types. The Importance of Electric Field Distribution in ECCT In Electro-Capacitive Cancer Therapy (ECCT), the distribution and intensity of electric fields (EF) play a crucial role in determining the therapy’s effectiveness. The success of ECCT in inhibiting cancer cell growth relies on the precise application of electric fields, which disrupt critical cellular processes such as mitosis. Mitosis, the process through which cancer cells divide, is highly sensitive to external electric forces. By carefully controlling the intensity and distribution of electric fields, ECCT can interfere with mitotic spindle formation, leading to failed cell division and, ultimately, cancer cell death [4]. EF intensity is critical because different tissues and cells respond variably to electric fields. Cancer cells, due to their irregular growth, have altered electrical properties, including differences in membrane potential and conductivity, which make them more susceptible to electric field interference. During mitosis, when cancer cells are actively dividing, they are particularly vulnerable to changes in their electrical environment. A carefully calibrated electric field can destabilize the mitotic spindle apparatus, causing the division process to stall or fail. Without proper cell division, cancer cells either die or become nonviable, slowing tumor growth and spread. To achieve optimal results, electric field intensity must be carefully managed to ensure that it reaches the cancerous tissue with enough strength to disrupt mitosis. At the same time, the surrounding healthy tissue, which has different electrical properties, should remain largely unaffected. The key is finding the right balance: fields that are strong enough to halt cancer cell proliferation while being weak enough to avoid harming normal cells. Previous studies have shown that the selective nature of EF intensity in ECCT allows for this precision. For example, research has demonstrated that electric fields at intermediate frequencies preferentially affect cancer cells due to their abnormal electrical activities. These fields can target cancer cells by exploiting the differences in dielectric properties
Revolutionizing Cancer Treatment: How Innovative Technology Powers Electro-Capacitive Cancer Therapy
Electro-Capacitive Cancer Therapy (ECCT) is emerging as a promising treatment method for cancer, offering a novel approach to combat tumor growth. Unlike traditional cancer therapies, which often involve invasive procedures, chemotherapy, or radiation, ECCT leverages the power of electric fields to target cancer cells. By applying alternating electric fields to the tumor site, ECCT aims to disrupt the cell division processes within cancer cells, thereby inhibiting their growth and promoting cell death. This innovative approach is garnering attention for its potential to enhance treatment outcomes while minimizing damage to surrounding healthy tissues [1]. The role of technology in enhancing the effectiveness of ECCT cannot be overstated. Advanced electronic components, such as frequency generators and amplifiers, are critical for creating the precise electric fields required for treatment. By optimizing these technologies, researchers can ensure that the electric fields generated are effective in penetrating tumor tissues while being safe for normal cells. The recent advancements in integrated circuit (IC) technology, particularly in 300nm CMOS (Complementary Metal-Oxide-Semiconductor) technology, have made it possible to develop more efficient and compact devices that can accurately control and deliver the necessary electrical signals for ECCT [2]. This technological progress not only boosts the efficacy of the therapy but also paves the way for its widespread adoption in clinical settings. Electro-Capacitive Cancer Therapy (ECCT) utilizes electric fields as a non-invasive approach to target and destroy cancer cells. The fundamental principle behind ECCT is that cancer cells exhibit distinct electrical properties compared to healthy cells. By applying controlled electric fields, ECCT can preferentially disrupt the physiological activities of cancer cells, particularly during the cell division process known as mitosis. This disruption leads to increased cell stress and ultimately induces apoptosis (programmed cell death), thereby reducing tumor size and growth. One of the standout features of ECCT is its non-invasive nature. This means that the therapy can be administered without the need for surgical procedures, which is particularly beneficial for patients who may be hesitant to undergo more traditional, invasive treatments. Instead of needing to enter the body, the electric fields can be applied externally, making the treatment more accessible and potentially reducing recovery times. Patients benefit from a lower risk of complications, less pain, and a more comfortable treatment experience. The non-invasive aspect of ECCT aligns with a growing trend in cancer treatment that emphasizes minimally invasive procedures that prioritize patient quality of life while still effectively managing cancer [3]. The Role of Frequency Generators and Amplifiers Frequency generators and amplifiers are essential components of the Electro-Capacitive Cancer Therapy (ECCT) system. These devices play a pivotal role in generating the specific electric signals required to effectively target and treat cancer cells. Frequency generators produce alternating electric fields by generating signals at precise frequencies and voltage levels. In the context of ECCT, the frequency of these signals is crucial, as different frequencies can have varying effects on cellular activities. For instance, certain frequencies may be more effective in disrupting the mitotic process in cancer cells, thereby enhancing the therapeutic efficacy of the treatment. By adjusting the frequency, researchers can optimize the therapy to maximize its impact on tumor growth while minimizing side effects on surrounding healthy tissues. Amplifiers complement the function of frequency generators by boosting the generated signals to the required power levels for effective treatment delivery. They ensure that the electric fields can penetrate tumor tissues adequately, providing the necessary strength to disrupt cancer cell proliferation. Together, these components allow for precise control over the electric fields used in ECCT, ensuring that they are tailored to the specific requirements of the treatment [4]. Designing the Technology The design process of the frequency generator and amplifier level converter is a crucial step in developing an effective ECCT system. This process begins with understanding the standard operation mode of ECCT, which involves specifying the input and output signal parameters, including voltage, form, and frequency. The design must ensure that the generated signals align with the therapeutic needs for optimal efficacy. The use of 300nm CMOS technology plays a significant role in this design process. CMOS (Complementary Metal-Oxide-Semiconductor) technology is renowned for its low power consumption and high efficiency, making it an excellent choice for developing electronic components for medical applications. The 300nm process node allows for miniaturization of the circuit elements, which leads to smaller, more efficient devices that can be integrated into portable ECCT systems. This technology not only facilitates the creation of complex circuit designs but also enhances the performance and reliability of the frequency generators and amplifiers [5]. By utilizing 300nm CMOS technology, researchers can develop frequency generators and amplifiers that meet the stringent demands of ECCT, including high signal fidelity, low noise levels, and effective signal amplification. This integration of advanced technology is essential for realizing the full potential of ECCT as a cutting-edge cancer treatment modality, paving the way for future innovations in cancer therapy. The study on the design of frequency generators and amplifiers for Electro-Capacitive Cancer Therapy (ECCT) revealed significant results that underscore the technology’s potential. One of the main findings was the capability of the developed system to produce a specific output voltage of 19Vpp (volts peak-to-peak) and a frequency of 100 kHz. These specifications are critical for the effective operation of ECCT, as they align with the required parameters necessary to create the electric fields that disrupt cancer cell division [6]. The ability to generate 19Vpp ensures that the electric fields are sufficiently strong to penetrate tumor tissues, while the 100 kHz frequency is particularly effective in targeting the abnormal electrical activities of cancer cells. This combination of voltage and frequency allows for optimal disruption of cellular processes, enhancing the therapy’s ability to inhibit tumor growth while minimizing harm to surrounding healthy cells. The study confirms that the integrated design meets the rigorous demands of ECCT operation, providing a solid foundation for future applications in cancer treatment. The technological advancements demonstrated in this study have far-reaching implications for cancer treatment. By optimizing the design of frequency generators and amplifiers, researchers
How Electro-Capacitive Therapy May Halt Cancer: Exploring the Role of CCL2 and IL18 in Tumor Suppression
Electro-Capacitive Cancer Therapy (ECCT) is a cutting-edge, non-invasive treatment that harnesses the power of alternating electric fields to target and disrupt cancer cell proliferation. Unlike conventional treatments like chemotherapy or radiation, ECCT offers a novel approach by applying external electric fields to tumors, which may selectively interfere with cancer cell division while leaving healthy cells relatively unaffected. The growing interest in ECCT stems from its potential to inhibit tumor growth without causing the widespread damage typically seen with traditional cancer treatments. Studies have shown that ECCT can reduce tumor proliferation by inducing apoptosis (programmed cell death) and inhibiting key signaling pathways involved in cancer growth. In addition, ECCT’s non-invasive nature makes it an attractive option for patients seeking alternatives to more aggressive therapies [1]. How ECCT works is based on the application of alternating current electric fields (AC-EF) between two capacitive electrodes positioned near the tumor. These electric fields are applied at specific frequencies and intensities designed to exploit the unique electrical properties of cancer cells. Cancer cells have different dielectric properties compared to normal cells, particularly during mitosis (cell division). By applying alternating electric fields at intermediate frequencies, ECCT can destabilize the cancer cells’ membrane integrity, interrupting mitosis and leading to cell death. This effect is particularly important in slowing tumor growth and preventing metastasis. Role of CCL2 and IL18 in Cancer In the context of cancer progression, Cytokines such as CCL2 and IL18 play critical roles in promoting inflammation and influencing tumor behavior. These proteins are essential for cell signaling, and their overexpression can lead to the proliferation, migration, and survival of cancer cells. Understanding how these cytokines interact with ECCT is key to grasping the therapeutic potential of this treatment [2]. CCL2: A Key Player in Cancer Metastasis CCL2 (C-C motif chemokine ligand 2) is a chemokine that is heavily involved in the recruitment of monocytes to sites of inflammation and injury. In the context of cancer, it is known to promote metastasis, particularly in breast cancer. CCL2 aids in the migration of immune cells, such as macrophages, to the tumor microenvironment, where they can support tumor growth and metastasis by enhancing the inflammatory response. It also promotes angiogenesis (the formation of new blood vessels), which tumors rely on for nutrients and oxygen. In breast cancer, CCL2 has been associated with a higher propensity for metastatic spread, especially to distant organs like the lungs. This makes it a crucial target for therapies aiming to control tumor growth and metastasis. By reducing CCL2 expression, treatments like ECCT may help suppress the tumor’s ability to grow and invade other tissues [3]. IL18: Modulating the Tumor Immune Microenvironment Interleukin-18 (IL18) is another cytokine that plays a significant role in cancer progression, particularly in modulating the immune response. It is known to influence the activity of macrophages, a type of immune cell that can either promote or inhibit tumor growth depending on the signals they receive. IL18 has been shown to facilitate a pro-tumor immune environment by promoting macrophage polarization towards a tumor-supportive phenotype, which contributes to excessive angiogenesis—the formation of new blood vessels that feed tumor cells. IL18 can drive inflammation in the tumor microenvironment, leading to enhanced tumor survival and growth. By increasing macrophage activity and angiogenesis, IL18 essentially creates conditions that allow tumors to thrive. In the context of breast cancer, IL18 has been linked to more aggressive tumor behavior and poorer prognosis [4]. In studies involving ECCT, it has been observed that exposure to electric fields may downregulate the expression of IL18, thus reducing its ability to promote tumor growth and angiogenesis. This suggests that one of ECCT’s anti-tumor mechanisms may involve suppressing the inflammatory and immune-modulating effects of cytokines like IL18, which are otherwise supportive of cancer progression. Together, CCL2 and IL18 represent critical targets in cancer therapy, particularly in treatments aimed at controlling tumor growth and metastasis. By regulating the expression of these cytokines, therapies such as ECCT may not only halt cancer proliferation but also modulate the immune environment to be less favorable for cancer development. This dual action direct inhibition of cancer cells and modification of the tumor microenvironment makes ECCT a promising approach in the fight against cancer [5]. The Experiment: How ECCT Affects Breast Tumor Growth In this study, researchers investigated how Noncontact Electro-Capacitive Cancer Therapy (ECCT) impacts breast tumor growth in a controlled experiment using rats. The animal model involved inducing breast tumors in rats with a carcinogen known as 7,12-dimethylbenz[a]anthracene (DMBA), which reliably causes breast cancer in rodents, making it a suitable model for studying human breast cancer. The experiment was designed with four distinct groups of rats: NINT (Non-induced, no treatment): Rats that were not exposed to DMBA and did not receive ECCT treatment. This served as the healthy control group. NIT (Non-induced, treatment): Rats that were not exposed to DMBA but were treated with ECCT. This group helped to observe any effects of ECCT on non-cancerous tissue. INT (Induced, no treatment): Rats that were induced with breast tumors via DMBA but did not receive ECCT. This group allowed for the observation of the natural progression of tumor growth. IT (Induced, treatment): Rats that were induced with breast tumors via DMBA and treated with ECCT. This group was the focus of the study, allowing the researchers to observe how ECCT influences tumor growth. For the ECCT treatment, an alternating current electric field (AC-EF) of 18 Vpp (low intensity) and 150 kHz (intermediate frequency) was applied between two capacitive electrodes placed around the rat cage. This created an external electric field that acted on the tumors without direct contact with the animals. The aim was to evaluate how the electric field influences tumor behavior over time [6]. To assess tumor growth and the biological effects of ECCT, researchers analyzed specific biomarkers that are commonly used to track cancer activity: PCNA (Proliferating Cell Nuclear Antigen): A marker of cell proliferation, which indicates the rate at which cancer cells are dividing and growing. ErbB2 (also known as HER2): A